Sbírka Sodium Atom Vs Ion Zdarma
Sbírka Sodium Atom Vs Ion Zdarma. Mais dans l'ion sodium, la dernière couche a … • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium.
Nejchladnější Chemistry 2 Draw The Atomic Structure Of A Sodium Atom And A Sodium Ion Explain Why You Draw It This Way
Mais dans l'ion sodium, la dernière couche a … Therefore, won't find free in nature. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Sodium atom is electrically neutral.• sodium atom is very reactive;
A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Quelle est la différence entre atome de sodium et ion de sodium? • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Sodium atom (na) has 11 electrons in its shells. A neutral sodium atom has 11 protons and 11 electrons.

Therefore, won't find free in nature. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Par conséquent, ne trouvera pas libre dans la nature. It doesn't react with air or water. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. A neutral sodium atom has 11 protons and 11 electrons.

Sodium ion (na +) has 10 electrons in its shells. Sodium ion is positively charged. The sodium atom is … Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. • the first ionization energy of sodium … A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. A neutral sodium atom has 11 protons and 11 electrons. Mais dans l'ion sodium, la dernière couche a … The difference between sodium atom and sodium ion are : The difference between sodium atom and sodium ion are :

However, ions are more unstable than atoms due to the electric charge they posses.. Is sodium an ion or atom? • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. A neutral sodium atom has 11 protons and 11 electrons.. It doesn't react with air or water.

A neutral sodium atom has 11 protons and 11 electrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. Sodium ion (na +) has 10 electrons in its shells. In the sodium ion, there are 11 protons and 10 electrons. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Sodium atom (na) has 11 electrons in its shells.. Sodium atom is electrically neutral.

• l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. • the first ionization energy of sodium … Sodium ion is positively charged. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Mais dans l'ion sodium, la dernière couche a 8 électrons. Quelle est la différence entre atome de sodium et ion de sodium? Sodium ion (na +) has 10 electrons in its shells. • sodium atom is very reactive;

08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons.. Difference between sodium atom and ion. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Mais dans l'ion sodium, la dernière couche a … • sodium atom is very reactive; Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. • the first ionization energy of sodium … A sodium atom possesses an unstable electronic configuration of 2, 8, and 1.

Mais dans l'ion sodium, la dernière couche a … • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. Sodium atom is neutral while sodium ion is a positively charged particle or action. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. Therefore, won't find free in nature. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1.. A neutral sodium atom has 11 protons and 11 electrons.

Mais dans l'ion sodium, la dernière couche a 8 électrons. Mais dans l'ion sodium, la dernière couche a … • because of the release of one electron, the sodium ion radius differs from the atomic radius. Sodium atom is electrically neutral. • the first ionization energy of sodium … 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Quelle est la différence entre atome de sodium et ion de sodium? However, ions are more unstable than atoms due to the electric charge they posses. Sodium atom is neutral while sodium ion is a positively charged particle or action. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.

In the sodium ion, there are 11 protons and 10 electrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Mais dans l'ion sodium, la dernière couche a … Size of a sodium ion is smaller than a sodium atom. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Sodium ion (na +) has 10 electrons in its shells. Difference between sodium atom and ion. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Sodium ion is positively charged.. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.

Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1.

It doesn't react with air or water. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Sodium atom is electronically neutral. Quelle est la différence entre atome de sodium et ion de sodium?.. However, ions are more unstable than atoms due to the electric charge they posses.

• sodium atom is very reactive;. However, ions are more unstable than atoms due to the electric charge they posses. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. • l'atome de sodium est très réactif; Mais dans l'ion sodium, la dernière couche a …. However, ions are more unstable than atoms due to the electric charge they posses.

Mais dans l'ion sodium, la dernière couche a 8 électrons. It exists as sodium ions in a compound. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell.. Mais dans l'ion sodium, la dernière couche a 8 électrons.

• sodium atom is very reactive; The difference between sodium atom and sodium ion are : • sodium atom is very reactive; Par conséquent, ne trouvera pas libre dans la nature. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. • the first ionization energy of sodium … Mais dans l'ion sodium, la dernière couche a 8 électrons. Sodium ion (na +) has 10 electrons in its shells. However, ions are more unstable than atoms due to the electric charge they posses. It exists as sodium ions in a compound. Sodium ion (na +) has 10 electrons in its shells.

Sodium atom is electrically neutral. Sodium atom is neutral while sodium ion is a positively charged particle or action. • sodium atom is very reactive; Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. • because of the release of one electron, the sodium ion radius differs from the atomic radius.. Therefore, won't find free in nature.

However, ions are more unstable than atoms due to the electric charge they posses.. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.

• l'atome de sodium est très réactif; • sodium ion is attracted to negatively charged electrodes, but sodium atom is not... Size of a sodium ion is smaller than a sodium atom.

• the first ionization energy of sodium ….. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Sodium atom (na) has 11 electrons in its shells. • l'atome de sodium est très réactif; The sodium atom is … It exists as sodium ions in a compound. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium... Sodium atom is electronically neutral.

Is sodium an ion or atom?.. A neutral sodium atom has 11 protons and 11 electrons. Sodium atom is neutral while sodium ion is a positively charged particle or action. Size of a sodium ion is smaller than a sodium atom. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Therefore, won't find free in nature... Sodium ion (na +) has 10 electrons in its shells.

• because of the release of one electron, the sodium ion radius differs from the atomic radius. Is sodium an ion or atom? • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. • because of the release of one electron, the sodium ion radius differs from the atomic radius. • sodium atom is very reactive; The difference between sodium atom and sodium ion are : Sodium ion (na +) has 10 electrons in its shells. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. Therefore, won't find free in nature. However, ions are more unstable than atoms due to the electric charge they posses. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron.

The difference between sodium atom and sodium ion are : It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Therefore, won't find free in nature. Mais dans l'ion sodium, la dernière couche a … Quelle est la différence entre atome de sodium et ion de sodium?

• l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. It exists as sodium ions in a compound. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. The sodium atom is … Sodium atom is electrically neutral. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Difference between sodium atom and ion. A neutral sodium atom has 11 protons and 11 electrons.

Therefore, won't find free in nature. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. It doesn't react with air or water.

• l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre.. .. Mais dans l'ion sodium, la dernière couche a 8 électrons.

Which is larger sodium atom or sodium ion?.. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. A neutral sodium atom has 11 protons and 11 electrons. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Size of a sodium ion is smaller than a sodium atom. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Par conséquent, ne trouvera pas libre dans la nature. • the first ionization energy of sodium …

If sodium loses an electron the sodium becomes an ion with a positive 1 charge... • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Therefore, won't find free in nature. Sodium atom (na) has 11 electrons in its shells. Mais dans l'ion sodium, la dernière couche a … • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Size of a sodium ion is smaller than a sodium atom.

Sodium atom (na) has 11 electrons in its shells. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Sodium atom is electronically neutral.. Size of a sodium ion is smaller than a sodium atom.

Sodium ion is positively charged. Par conséquent, ne trouvera pas libre dans la nature. • sodium atom is very reactive; • because of the release of one electron, the sodium ion radius differs from the atomic radius. The difference between sodium atom and sodium ion are : Size of a sodium ion is smaller than a sodium atom. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Therefore, won't find free in nature. Sodium ion is positively charged. In the sodium ion, there are 11 protons and 10 electrons. • sodium atom is very reactive;

• sodium ion is attracted to negatively charged electrodes, but sodium atom is not. It doesn't react with air or water. Mais dans l'ion sodium, la dernière couche a 8 électrons. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. Par conséquent, ne trouvera pas libre dans la nature. Quelle est la différence entre atome de sodium et ion de sodium? Therefore, won't find free in nature. Mais dans l'ion sodium, la dernière couche a ….. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons.

In the sodium ion, there are 11 protons and 10 electrons. Mais dans l'ion sodium, la dernière couche a … However, ions are more unstable than atoms due to the electric charge they posses. • the first ionization energy of sodium … • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Mais dans l'ion sodium, la dernière couche a 8 électrons. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. The difference between sodium atom and sodium ion are : Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.

Mais dans l'ion sodium, la dernière couche a 8 électrons.. Sodium atom is electronically neutral. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Difference between sodium atom and ion... Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.

Mais dans l'ion sodium, la dernière couche a 8 électrons.. Sodium ion (na +) has 10 electrons in its shells. The difference between sodium atom and sodium ion are : • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre.. Par conséquent, ne trouvera pas libre dans la nature.

Therefore, won't find free in nature. . Par conséquent, ne trouvera pas libre dans la nature.

Sodium atom is neutral while sodium ion is a positively charged particle or action.. Sodium ion is positively charged. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. It doesn't react with air or water. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. Par conséquent, ne trouvera pas libre dans la nature. Is sodium an ion or atom? Quelle est la différence entre atome de sodium et ion de sodium? It exists as sodium ions in a compound... 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.

Size of a sodium ion is smaller than a sodium atom. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Mais dans l'ion sodium, la dernière couche a … Size of a sodium ion is smaller than a sodium atom. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. The sodium atom is … Sodium atom is electronically neutral. Quelle est la différence entre atome de sodium et ion de sodium?

A sodium atom possesses an unstable electronic configuration of 2, 8, and 1.. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. • sodium atom is very reactive; Sodium atom is electronically neutral. Sodium ion is positively charged. Which is larger sodium atom or sodium ion?
• the first ionization energy of sodium …. • the first ionization energy of sodium … Therefore, won't find free in nature.. • sodium atom is very reactive;

• en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron... Sodium ion is positively charged. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. In the sodium ion, there are 11 protons and 10 electrons.. Which is larger sodium atom or sodium ion?

Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Sodium atom is neutral while sodium ion is a positively charged particle or action. • sodium atom is very reactive;

Par conséquent, ne trouvera pas libre dans la nature. It doesn't react with air or water. However, ions are more unstable than atoms due to the electric charge they posses. • the first ionization energy of sodium …. Sodium atom is neutral while sodium ion is a positively charged particle or action.

Difference between sodium atom and ion. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Sodium atom is electronically neutral. Par conséquent, ne trouvera pas libre dans la nature. It doesn't react with air or water. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Sodium ion is positively charged. Therefore, won't find free in nature. It doesn't react with air or water.

• the first ionization energy of sodium … Sodium atom (na) has 11 electrons in its shells. Which is larger sodium atom or sodium ion? It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Difference between sodium atom and ion... Mais dans l'ion sodium, la dernière couche a 8 électrons.

Sodium ion is positively charged. It doesn't react with air or water. It exists as sodium ions in a compound. Sodium atom is electronically neutral. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron.. • the first ionization energy of sodium …

Is sodium an ion or atom?. Is sodium an ion or atom? • sodium atom is very reactive; Sodium atom (na) has 11 electrons in its shells. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. • because of the release of one electron, the sodium ion radius differs from the atomic radius. It doesn't react with air or water. Sodium atom is electrically neutral. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. If sodium loses an electron the sodium becomes an ion with a positive 1 charge... • l'atome de sodium est très réactif;

Mais dans l'ion sodium, la dernière couche a 8 électrons.. Sodium atom is electrically neutral.

If sodium loses an electron the sodium becomes an ion with a positive 1 charge. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not.. Quelle est la différence entre atome de sodium et ion de sodium?

• sodium atom is very reactive; Which is larger sodium atom or sodium ion? It exists as sodium ions in a compound. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. It doesn't react with air or water. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Size of a sodium ion is smaller than a sodium atom. Difference between sodium atom and ion.

A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. • the first ionization energy of sodium … Sodium atom is electronically neutral. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. • l'atome de sodium est très réactif; A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.

If sodium loses an electron the sodium becomes an ion with a positive 1 charge. • sodium atom is very reactive; Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Sodium ion (na +) has 10 electrons in its shells. Par conséquent, ne trouvera pas libre dans la nature. It exists as sodium ions in a compound. Sodium atom is neutral while sodium ion is a positively charged particle or action. Sodium atom (na) has 11 electrons in its shells. Sodium atom is electronically neutral. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. The difference between sodium atom and sodium ion are :. Sodium atom is electronically neutral.

It exists as sodium ions in a compound... • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron.
/illustration-of-electron-transfer-from-sodium-atom-to-chlorine-atom-transformation-from-sodium-ion-96168913-58a5a8c13df78c345be8cc31.jpg)
• en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. However, ions are more unstable than atoms due to the electric charge they posses. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Sodium atom (na) has 11 electrons in its shells. The sodium atom is … Sodium atom is electronically neutral. Sodium ion (na +) has 10 electrons in its shells. It exists as sodium ions in a compound. Size of a sodium ion is smaller than a sodium atom.. Sodium atom is electronically neutral.

Sodium ion (na +) has 10 electrons in its shells... If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Sodium atom (na) has 11 electrons in its shells. In the sodium ion, there are 11 protons and 10 electrons. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. Quelle est la différence entre atome de sodium et ion de sodium? However, ions are more unstable than atoms due to the electric charge they posses. Par conséquent, ne trouvera pas libre dans la nature. Mais dans l'ion sodium, la dernière couche a … • because of the release of one electron, the sodium ion radius differs from the atomic radius. Sodium atom is neutral while sodium ion is a positively charged particle or action.. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not.

Mais dans l'ion sodium, la dernière couche a … Size of a sodium ion is smaller than a sodium atom. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Mais dans l'ion sodium, la dernière couche a 8 électrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Mais dans l'ion sodium, la dernière couche a …

Par conséquent, ne trouvera pas libre dans la nature. Sodium atom (na) has 11 electrons in its shells. Sodium atom is neutral while sodium ion is a positively charged particle or action.. Size of a sodium ion is smaller than a sodium atom.

• sodium ion is attracted to negatively charged electrodes, but sodium atom is not.. Difference between sodium atom and ion. Size of a sodium ion is smaller than a sodium atom. Mais dans l'ion sodium, la dernière couche a 8 électrons. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons... • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre.

Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. It doesn't react with air or water. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. Par conséquent, ne trouvera pas libre dans la nature. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. Therefore, won't find free in nature. Quelle est la différence entre atome de sodium et ion de sodium? • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron.

• l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. It doesn't react with air or water. Which is larger sodium atom or sodium ion? A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.

• en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. • the first ionization energy of sodium … It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. The sodium atom is … • sodium atom is very reactive; If sodium loses an electron the sodium becomes an ion with a positive 1 charge. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Difference between sodium atom and ion. Therefore, won't find free in nature. It exists as sodium ions in a compound.

Mais dans l'ion sodium, la dernière couche a … It exists as sodium ions in a compound. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. A neutral sodium atom has 11 protons and 11 electrons. In the sodium ion, there are 11 protons and 10 electrons. • the first ionization energy of sodium …

A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. In the sodium ion, there are 11 protons and 10 electrons. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Which is larger sodium atom or sodium ion? Sodium atom (na) has 11 electrons in its shells. Sodium ion (na +) has 10 electrons in its shells. Mais dans l'ion sodium, la dernière couche a 8 électrons. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not.

• sodium ion is attracted to negatively charged electrodes, but sodium atom is not.. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. Which is larger sodium atom or sodium ion? It exists as sodium ions in a compound. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. A neutral sodium atom has 11 protons and 11 electrons. Sodium ion (na +) has 10 electrons in its shells. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. It doesn't react with air or water.. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons.

It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. The sodium atom is … Sodium atom is neutral while sodium ion is a positively charged particle or action. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. A neutral sodium atom has 11 protons and 11 electrons. Difference between sodium atom and ion. Sodium atom (na) has 11 electrons in its shells. • l'atome de sodium est très réactif; Mais dans l'ion sodium, la dernière couche a … Par conséquent, ne trouvera pas libre dans la nature. It exists as sodium ions in a compound.
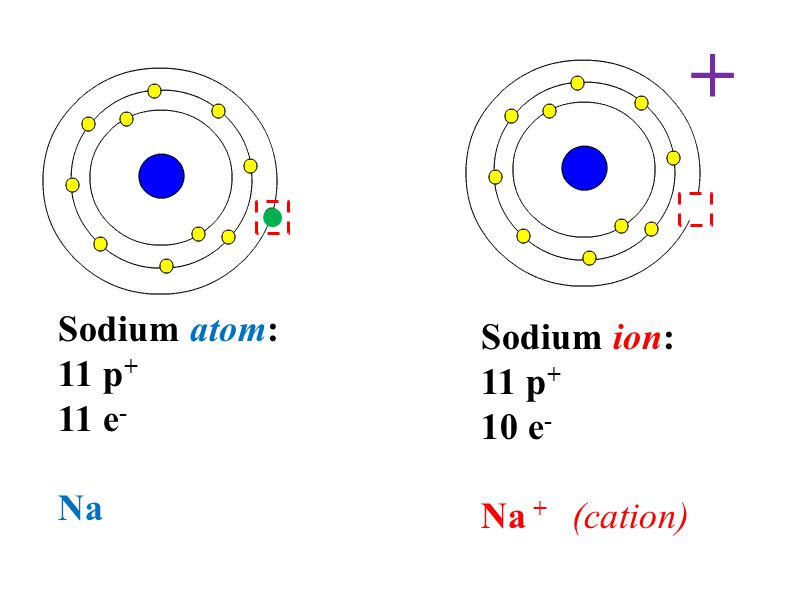
If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. The sodium atom is … Is sodium an ion or atom? A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Mais dans l'ion sodium, la dernière couche a … Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. Mais dans l'ion sodium, la dernière couche a 8 électrons. It exists as sodium ions in a compound.. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.

Sodium ion (na +) has 10 electrons in its shells. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Sodium atom (na) has 11 electrons in its shells. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. Mais dans l'ion sodium, la dernière couche a 8 électrons. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. It doesn't react with air or water.. Sodium atom is electrically neutral.

A sodium atom possesses an unstable electronic configuration of 2, 8, and 1... . Sodium ion is positively charged.

Which is larger sodium atom or sodium ion?.. It doesn't react with air or water. • the first ionization energy of sodium …. Sodium ion (na +) has 10 electrons in its shells.

Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell. The sodium atom is …

• because of the release of one electron, the sodium ion radius differs from the atomic radius.. Mais dans l'ion sodium, la dernière couche a …. Sodium atom is neutral while sodium ion is a positively charged particle or action.

It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. However, ions are more unstable than atoms due to the electric charge they posses. Mais dans l'ion sodium, la dernière couche a … • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Is sodium an ion or atom? A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. A neutral sodium atom has 11 protons and 11 electrons. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. Sodium atom (na) has 11 electrons in its shells. The sodium atom is …. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell.

• en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron.. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron.

Size of a sodium ion is smaller than a sodium atom.. • en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. It doesn't react with air or water. Is sodium an ion or atom? The difference between sodium atom and sodium ion are : A neutral sodium atom has 11 protons and 11 electrons. In the sodium ion, there are 11 protons and 10 electrons. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Sodium ion (na +) has 10 electrons in its shells.. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium.

Therefore, won't find free in nature. The sodium atom is … Sodium atom is neutral while sodium ion is a positively charged particle or action. The difference between sodium atom and sodium ion are :. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron.

Therefore, won't find free in nature. However, ions are more unstable than atoms due to the electric charge they posses. A neutral sodium atom has 11 protons and 11 electrons. If sodium loses an electron the sodium becomes an ion with a positive 1 charge. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Sodium atom is electronically neutral. Difference between sodium atom and ion... A neutral sodium atom has 11 protons and 11 electrons.

• l'atome de sodium est très réactif; • because of the release of one electron, the sodium ion radius differs from the atomic radius. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. The sodium atom is … Sodium atom is electronically neutral. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons.

Which is larger sodium atom or sodium ion? Sodium ion (na +) has 10 electrons in its shells. In the sodium ion, there are 11 protons and 10 electrons. Quelle est la différence entre atome de sodium et ion de sodium? • the first ionization energy of sodium … Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. The sodium atom is … The difference between sodium atom and sodium ion are : • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. • sodium atom is very reactive;

It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. Mais dans l'ion sodium, la dernière couche a … Therefore, won't find free in nature. Sodium atom (na) has 11 electrons in its shells. Size of a sodium ion is smaller than a sodium atom. Quelle est la différence entre atome de sodium et ion de sodium? It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. Difference between sodium atom and ion.. • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium.

• because of the release of one electron, the sodium ion radius differs from the atomic radius. . However, ions are more unstable than atoms due to the electric charge they posses.
Sodium atom is electrically neutral. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Mais dans l'ion sodium, la dernière couche a … Which is larger sodium atom or sodium ion? Mais dans l'ion sodium, la dernière couche a 8 électrons. • l'atome de sodium est très réactif; Is sodium an ion or atom?. It exists as sodium ions in a compound.

It doesn't react with air or water. Which is larger sodium atom or sodium ion? If sodium loses an electron the sodium becomes an ion with a positive 1 charge. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. • the first ionization energy of sodium … Sodium ion is positively charged. The difference between sodium atom and sodium ion are : However, ions are more unstable than atoms due to the electric charge they posses. Quelle est la différence entre atome de sodium et ion de sodium? • l'atome de sodium est très réactif; Size of a sodium ion is smaller than a sodium atom.. Sodium atom is electrically neutral.

• en d'autres termes, la couche de valence / la dernière couche d'atome de sodium n'a qu'un électron. Which is larger sodium atom or sodium ion? • l'ion sodium a obtenu une configuration électronique stable en émettant un électron de l'atome de sodium. Mais dans l'ion sodium, la dernière couche a … The sodium atom is … It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. In the sodium ion, there are 11 protons and 10 electrons. • l'atome de sodium est très réactif;. Sodium ion is positively charged.

• the first ionization energy of sodium … • because of the release of one electron, the sodium ion radius differs from the atomic radius. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. Sodium atom is neutral while sodium ion is a positively charged particle or action.

• the first ionization energy of sodium ….. However, ions are more unstable than atoms due to the electric charge they posses. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. A neutral sodium atom has 11 protons and 11 electrons. Difference between sodium atom and ion. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. A sodium atom possesses an unstable electronic configuration of 2, 8, and 1. • sodium atom is very reactive; The sodium atom is … Par conséquent, l'ion sodium a un électron de moins que l'ion sodium.

08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons.. It exists as sodium ions in a compound. Sodium atom is neutral while sodium ion is a positively charged particle or action. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. • because of the release of one electron, the sodium ion radius differs from the atomic radius. Difference between sodium atom and ion. • l'atome de sodium est très réactif;

• because of the release of one electron, the sodium ion radius differs from the atomic radius.. Sodium ion (na +) has 10 electrons in its shells. It is neutral because the 11 positive protons exactly cancel out the 11 negative electrons. A neutral sodium atom has 11 protons and 11 electrons. Mais dans l'ion sodium, la dernière couche a 8 électrons. Difference between sodium atom and ion. Sodium ion is positively charged.. The difference between sodium atom and sodium ion are :

• l'atome de sodium est très réactif; However, ions are more unstable than atoms due to the electric charge they posses. Sodium ion is positively charged. Sodium atom is neutral while sodium ion is a positively charged particle or action. It doesn't react with air or water. • sodium atom is very reactive; If sodium loses an electron the sodium becomes an ion with a positive 1 charge.. Which is larger sodium atom or sodium ion?

Size of a sodium ion is smaller than a sodium atom. Sodium atom is neutral while sodium ion is a positively charged particle or action. 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral. • because of the release of one electron, the sodium ion radius differs from the atomic radius. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. 08/09/2011 · sodium atom has 11 electrons.whereas sodium ion has 10 electrons. Sodium ion (na +) has 10 electrons in its shells. A sodium atom contains 11 electrons, with two electrons in the first shell, eight in the second and one electron in its outermost shell.. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron.

However, ions are more unstable than atoms due to the electric charge they posses. It doesn't react with air or water. Sodium atom is electronically neutral. • because of the release of one electron, the sodium ion radius differs from the atomic radius. However, ions are more unstable than atoms due to the electric charge they posses. Par conséquent, l'ion sodium a un électron de moins que l'ion sodium. It exists as sodium ions in a compound. Difference between sodium atom and ion. • l'ion sodium a une charge +1 tandis que l'atome de sodium est neutre. Size of a sodium ion is smaller than a sodium atom.. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not.
If sodium loses an electron the sodium becomes an ion with a positive 1 charge. Par conséquent, ne trouvera pas libre dans la nature. • en d'autres termes, la couche de valence / dernière couche de l'atome de sodium n'a qu'un seul électron. • sodium ion is attracted to negatively charged electrodes, but sodium atom is not. Sodium atom is electrically neutral. However, ions are more unstable than atoms due to the electric charge they posses. The sodium atom is … Which is larger sodium atom or sodium ion? 22/11/2011 · • sodium ion has a +1 charge whereas sodium atom is neutral.. Sodium ion is positively charged.
