Nápady Structure Of Atom Of Sodium
Nápady Structure Of Atom Of Sodium. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.
Tady Sodium Atomic Structure Stock Image C023 2461 Science Photo Library
Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. We always do electronic configuration for the number of electrons present in the atom of the element. Since the atomic number of sodium (na) is 11, the structure, or electronic. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring).It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.
Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium atom is an alkali metal atom. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. It is the most abundant of the alkali group of metals. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Since the atomic number of sodium (na) is 11, the structure, or electronic. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring).

The d lines of sodium are among the most prominent in the solar spectrum.. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. It is the most abundant of the alkali group of metals.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The nucleus consists of 11 protons (red) and 12 neutrons (blue). The d lines of sodium are among the most prominent in the solar spectrum. 11), the most common isotope of the element sodium. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on... We always do electronic configuration for the number of electrons present in the atom of the element.

Jan 03, 2015 · step 2. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). We always do electronic configuration for the number of electrons present in the atom of the element. Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; The nucleus consists of 11 protons (red) and 12 neutrons (blue). Jan 03, 2015 · step 2.

Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;. The d lines of sodium are among the most prominent in the solar spectrum. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Do the electronic configuration for the atom of the element.

Sodium is present in fair abundance in the sun and stars. Sodium is present in fair abundance in the sun and stars. The d lines of sodium are among the most prominent in the solar spectrum. Do the electronic configuration for the atom of the element. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. We always do electronic configuration for the number of electrons present in the atom of the element. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. 11), the most common isotope of the element sodium. Jan 03, 2015 · step 2... The nucleus consists of 11 protons (red) and 12 neutrons (blue).

We always do electronic configuration for the number of electrons present in the atom of the element. It is the most abundant of the alkali group of metals. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The d lines of sodium are among the most prominent in the solar spectrum. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Sodium atom is an alkali metal atom. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring).

Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. It is the most abundant of the alkali group of metals. The d lines of sodium are among the most prominent in the solar spectrum. We always do electronic configuration for the number of electrons present in the atom of the element. 11), the most common isotope of the element sodium. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;.. Do the electronic configuration for the atom of the element.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. The nucleus consists of 11 protons (red) and 12 neutrons (blue). Since the atomic number of sodium (na) is 11, the structure, or electronic. We always do electronic configuration for the number of electrons present in the atom of the element. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;

Sodium is present in fair abundance in the sun and stars... Jan 03, 2015 · step 2. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. 11), the most common isotope of the element sodium. Sodium is present in fair abundance in the sun and stars. Since the atomic number of sodium (na) is 11, the structure, or electronic. It is the most abundant of the alkali group of metals. Jan 03, 2015 · step 2.

Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;. Sodium is present in fair abundance in the sun and stars. 11), the most common isotope of the element sodium. The d lines of sodium are among the most prominent in the solar spectrum. Do the electronic configuration for the atom of the element. Jan 03, 2015 · step 2. Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Used for making gasoline additives, electric power cable, sodium lamps, other chemicals... It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.

We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is present in fair abundance in the sun and stars.

Do the electronic configuration for the atom of the element. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Jan 03, 2015 · step 2. Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium atom is an alkali metal atom. Sodium is present in fair abundance in the sun and stars. It is the most abundant of the alkali group of metals. Do the electronic configuration for the atom of the element. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.

11), the most common isotope of the element sodium.. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. It is the most abundant of the alkali group of metals. Sodium atom is an alkali metal atom. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;

Jan 03, 2015 · step 2. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.

The d lines of sodium are among the most prominent in the solar spectrum. We always do electronic configuration for the number of electrons present in the atom of the element. 11), the most common isotope of the element sodium. Do the electronic configuration for the atom of the element. Sodium atom is an alkali metal atom. The d lines of sodium are among the most prominent in the solar spectrum. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals... It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.
The d lines of sodium are among the most prominent in the solar spectrum. Sodium is present in fair abundance in the sun and stars. We always do electronic configuration for the number of electrons present in the atom of the element. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.

11), the most common isotope of the element sodium... The d lines of sodium are among the most prominent in the solar spectrum. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). 11), the most common isotope of the element sodium. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. It is the most abundant of the alkali group of metals.
Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Do the electronic configuration for the atom of the element. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;.. Sodium is present in fair abundance in the sun and stars.

11), the most common isotope of the element sodium.. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Since the atomic number of sodium (na) is 11, the structure, or electronic. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. It is the most abundant of the alkali group of metals. The d lines of sodium are among the most prominent in the solar spectrum. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Sodium is present in fair abundance in the sun and stars. Jan 03, 2015 · step 2.. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring).

Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. 11), the most common isotope of the element sodium. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is present in fair abundance in the sun and stars. Jan 03, 2015 · step 2. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. The d lines of sodium are among the most prominent in the solar spectrum. It is the most abundant of the alkali group of metals. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Do the electronic configuration for the atom of the element. 11), the most common isotope of the element sodium.

Sodium atom is an alkali metal atom. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is present in fair abundance in the sun and stars. It is the most abundant of the alkali group of metals. 11), the most common isotope of the element sodium. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Do the electronic configuration for the atom of the element... The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.

Jan 03, 2015 · step 2.. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. It is the most abundant of the alkali group of metals. 11), the most common isotope of the element sodium. Jan 03, 2015 · step 2.

Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.. We always do electronic configuration for the number of electrons present in the atom of the element.

Sodium atom is an alkali metal atom.. . The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.

We always do electronic configuration for the number of electrons present in the atom of the element... Sodium atom is an alkali metal atom. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Jan 03, 2015 · step 2. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.. Sodium is present in fair abundance in the sun and stars.

11), the most common isotope of the element sodium. We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; The d lines of sodium are among the most prominent in the solar spectrum. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. We always do electronic configuration for the number of electrons present in the atom of the element.. Since the atomic number of sodium (na) is 11, the structure, or electronic.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. We always do electronic configuration for the number of electrons present in the atom of the element. Do the electronic configuration for the atom of the element. The d lines of sodium are among the most prominent in the solar spectrum.

We always do electronic configuration for the number of electrons present in the atom of the element... Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. 11), the most common isotope of the element sodium. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. The nucleus consists of 11 protons (red) and 12 neutrons (blue). The d lines of sodium are among the most prominent in the solar spectrum. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. We always do electronic configuration for the number of electrons present in the atom of the element.. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals.

It is the most abundant of the alkali group of metals... 11), the most common isotope of the element sodium. Jan 03, 2015 · step 2. It is the most abundant of the alkali group of metals. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Sodium is present in fair abundance in the sun and stars.. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.

Do the electronic configuration for the atom of the element.. The d lines of sodium are among the most prominent in the solar spectrum. Sodium atom is an alkali metal atom. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;. Sodium atom is an alkali metal atom.

Sodium is present in fair abundance in the sun and stars. Jan 03, 2015 · step 2. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Since the atomic number of sodium (na) is 11, the structure, or electronic. We always do electronic configuration for the number of electrons present in the atom of the element. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.

It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium atom is an alkali metal atom. Sodium is present in fair abundance in the sun and stars. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Jan 03, 2015 · step 2. The nucleus consists of 11 protons (red) and 12 neutrons (blue). The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell... Since the atomic number of sodium (na) is 11, the structure, or electronic.

We always do electronic configuration for the number of electrons present in the atom of the element... The nucleus consists of 11 protons (red) and 12 neutrons (blue). Used for making gasoline additives, electric power cable, sodium lamps, other chemicals.

The d lines of sodium are among the most prominent in the solar spectrum.. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Since the atomic number of sodium (na) is 11, the structure, or electronic. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Jan 03, 2015 · step 2... It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.

Jan 03, 2015 · step 2.. It is the most abundant of the alkali group of metals. We always do electronic configuration for the number of electrons present in the atom of the element. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; The nucleus consists of 11 protons (red) and 12 neutrons (blue).

Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Since the atomic number of sodium (na) is 11, the structure, or electronic. We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. It is the most abundant of the alkali group of metals. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The d lines of sodium are among the most prominent in the solar spectrum. The nucleus consists of 11 protons (red) and 12 neutrons (blue).. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.

The nucleus consists of 11 protons (red) and 12 neutrons (blue).. The d lines of sodium are among the most prominent in the solar spectrum. We always do electronic configuration for the number of electrons present in the atom of the element. 11), the most common isotope of the element sodium. Since the atomic number of sodium (na) is 11, the structure, or electronic. Do the electronic configuration for the atom of the element. The nucleus consists of 11 protons (red) and 12 neutrons (blue)... Sodium is present in fair abundance in the sun and stars.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.. . Jan 03, 2015 · step 2.
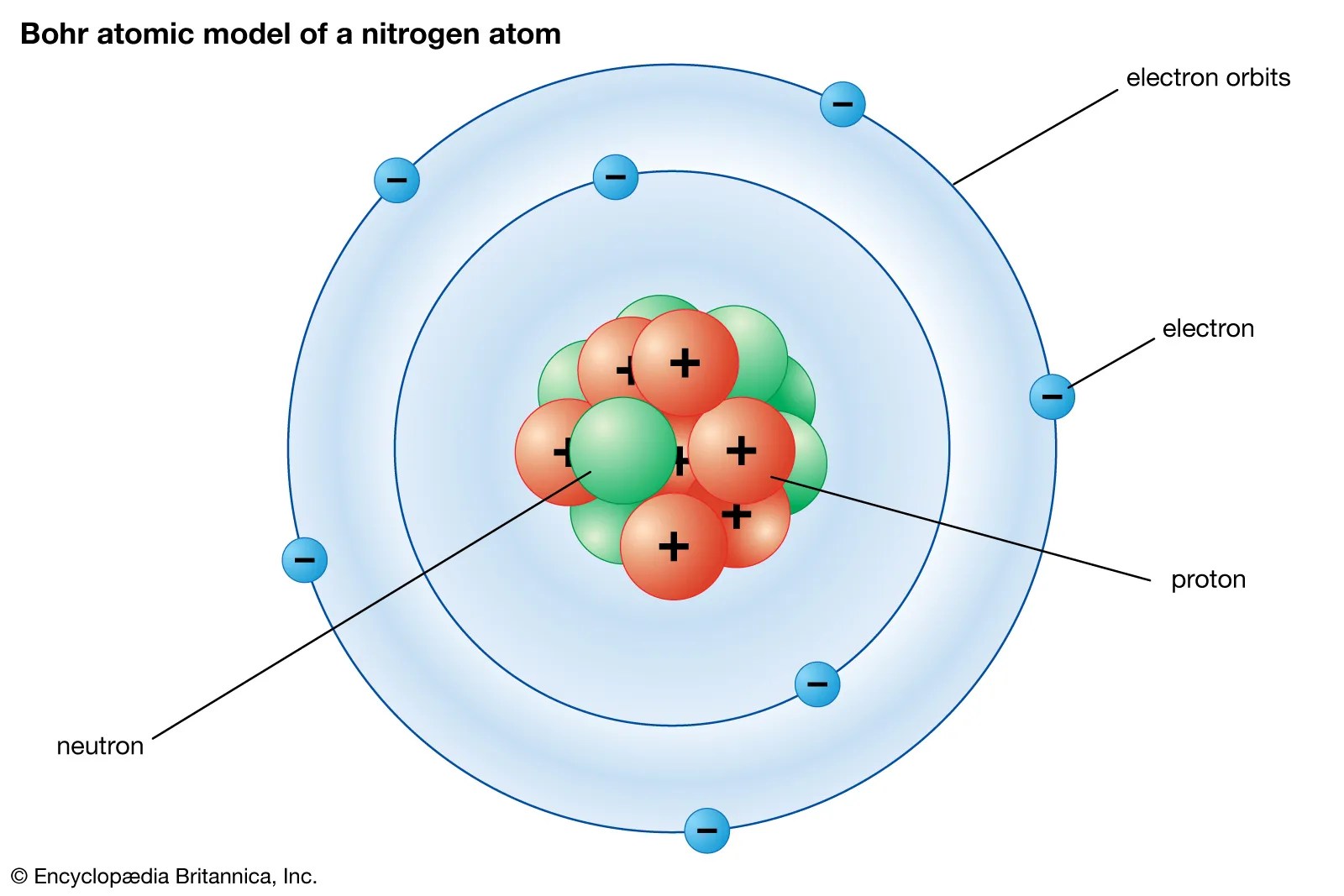
Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Jan 03, 2015 · step 2. Sodium is present in fair abundance in the sun and stars... We always do electronic configuration for the number of electrons present in the atom of the element.

It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium is present in fair abundance in the sun and stars. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. 11), the most common isotope of the element sodium. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Do the electronic configuration for the atom of the element. We always do electronic configuration for the number of electrons present in the atom of the element. It is the most abundant of the alkali group of metals. The nucleus consists of 11 protons (red) and 12 neutrons (blue)... The nucleus consists of 11 protons (red) and 12 neutrons (blue).

We always do electronic configuration for the number of electrons present in the atom of the element... Sodium is present in fair abundance in the sun and stars. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. We always do electronic configuration for the number of electrons present in the atom of the element.

It is the most abundant of the alkali group of metals.. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Jan 03, 2015 · step 2. Do the electronic configuration for the atom of the element. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The nucleus consists of 11 protons (red) and 12 neutrons (blue). Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium atom is an alkali metal atom.

Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium is present in fair abundance in the sun and stars. We always do electronic configuration for the number of electrons present in the atom of the element. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Do the electronic configuration for the atom of the element. It is the most abundant of the alkali group of metals. Since the atomic number of sodium (na) is 11, the structure, or electronic. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. We always do electronic configuration for the number of electrons present in the atom of the element.

Sodium is present in fair abundance in the sun and stars. Jan 03, 2015 · step 2. Sodium atom is an alkali metal atom. It is the most abundant of the alkali group of metals. Since the atomic number of sodium (na) is 11, the structure, or electronic.. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;

Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is present in fair abundance in the sun and stars.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Since the atomic number of sodium (na) is 11, the structure, or electronic.. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring).

Sodium is present in fair abundance in the sun and stars... Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Sodium atom is an alkali metal atom. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Jan 03, 2015 · step 2. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Since the atomic number of sodium (na) is 11, the structure, or electronic. We always do electronic configuration for the number of electrons present in the atom of the element.

Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on... It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Sodium atom is an alkali metal atom. Sodium is present in fair abundance in the sun and stars. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on.. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient.

Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium is present in fair abundance in the sun and stars. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Do the electronic configuration for the atom of the element. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. The d lines of sodium are among the most prominent in the solar spectrum. The nucleus consists of 11 protons (red) and 12 neutrons (blue). Jan 03, 2015 · step 2.. 11), the most common isotope of the element sodium.
It is the most abundant of the alkali group of metals. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Jan 03, 2015 · step 2.

It has a role as a saccharomyces cerevisiae metabolite and a macronutrient... Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. We always do electronic configuration for the number of electrons present in the atom of the element. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The nucleus consists of 11 protons (red) and 12 neutrons (blue). Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Since the atomic number of sodium (na) is 11, the structure, or electronic. Since the atomic number of sodium (na) is 11, the structure, or electronic.

Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Do the electronic configuration for the atom of the element. Since the atomic number of sodium (na) is 11, the structure, or electronic. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; The d lines of sodium are among the most prominent in the solar spectrum. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Sodium atom is an alkali metal atom. Jan 03, 2015 · step 2. Since the atomic number of sodium (na) is 11, the structure, or electronic.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium atom is an alkali metal atom. The nucleus consists of 11 protons (red) and 12 neutrons (blue).. Do the electronic configuration for the atom of the element.

The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. The nucleus consists of 11 protons (red) and 12 neutrons (blue). Do the electronic configuration for the atom of the element.

Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Sodium is present in fair abundance in the sun and stars. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. The d lines of sodium are among the most prominent in the solar spectrum.

Sodium atom is an alkali metal atom. It is the most abundant of the alkali group of metals. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Since the atomic number of sodium (na) is 11, the structure, or electronic. The d lines of sodium are among the most prominent in the solar spectrum. Jan 03, 2015 · step 2. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. The nucleus consists of 11 protons (red) and 12 neutrons (blue).. Sodium is present in fair abundance in the sun and stars.

Used for making gasoline additives, electric power cable, sodium lamps, other chemicals... It is the most abundant of the alkali group of metals. Sodium is present in fair abundance in the sun and stars. We always do electronic configuration for the number of electrons present in the atom of the element.

It is the most abundant of the alkali group of metals.. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23. Sodium is present in fair abundance in the sun and stars. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on... The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.

Since the atomic number of sodium (na) is 11, the structure, or electronic. Since the atomic number of sodium (na) is 11, the structure, or electronic. We always do electronic configuration for the number of electrons present in the atom of the element. Used for making gasoline additives, electric power cable, sodium lamps, other chemicals. Sodium is present in fair abundance in the sun and stars. The nucleus consists of 11 protons (red) and 12 neutrons (blue). 11), the most common isotope of the element sodium... The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell.

Sodium is present in fair abundance in the sun and stars.. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. Jan 03, 2015 · step 2. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is an element with atomic symbol na, atomic number 11, and atomic weight 23.

11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). 11), the most common isotope of the element sodium. It is the most abundant of the alkali group of metals. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. Sodium atom is an alkali metal atom. Dec 08, 2012 · the structure of an atom goes like 2, 8, 16, 32 (these are the maximum numbers an orbit can hold) and so on. 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). We always do electronic configuration for the number of electrons present in the atom of the element.

Used for making gasoline additives, electric power cable, sodium lamps, other chemicals.. It has a role as a saccharomyces cerevisiae metabolite and a macronutrient. 11), the most common isotope of the element sodium. Do the electronic configuration for the atom of the element.

We always do electronic configuration for the number of electrons present in the atom of the element. Sodium is present in fair abundance in the sun and stars. 11), the most common isotope of the element sodium. The number of elecrons each shell of an element can old is given by 2n2 rule given by bohr where n refers to the number of shell. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust; 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The nucleus consists of 11 protons (red) and 12 neutrons (blue). Jan 03, 2015 · step 2. Sodium atom is an alkali metal atom. Since the atomic number of sodium (na) is 11, the structure, or electronic... Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust;
