Ideje Atomic System Of Units Vynikající
Ideje Atomic System Of Units Vynikající. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.
Prezentováno Atomic Mass Unit Chemistry For Non Majors
× 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.
The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge... The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;
There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

× 10 7 c 2 farads per meter, or one statfarad per centimeter, … Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;.. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge... × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

× 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. . There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons... × 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.

× 10 7 c 2 farads per meter, or one statfarad per centimeter, ….. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;.. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.
The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;
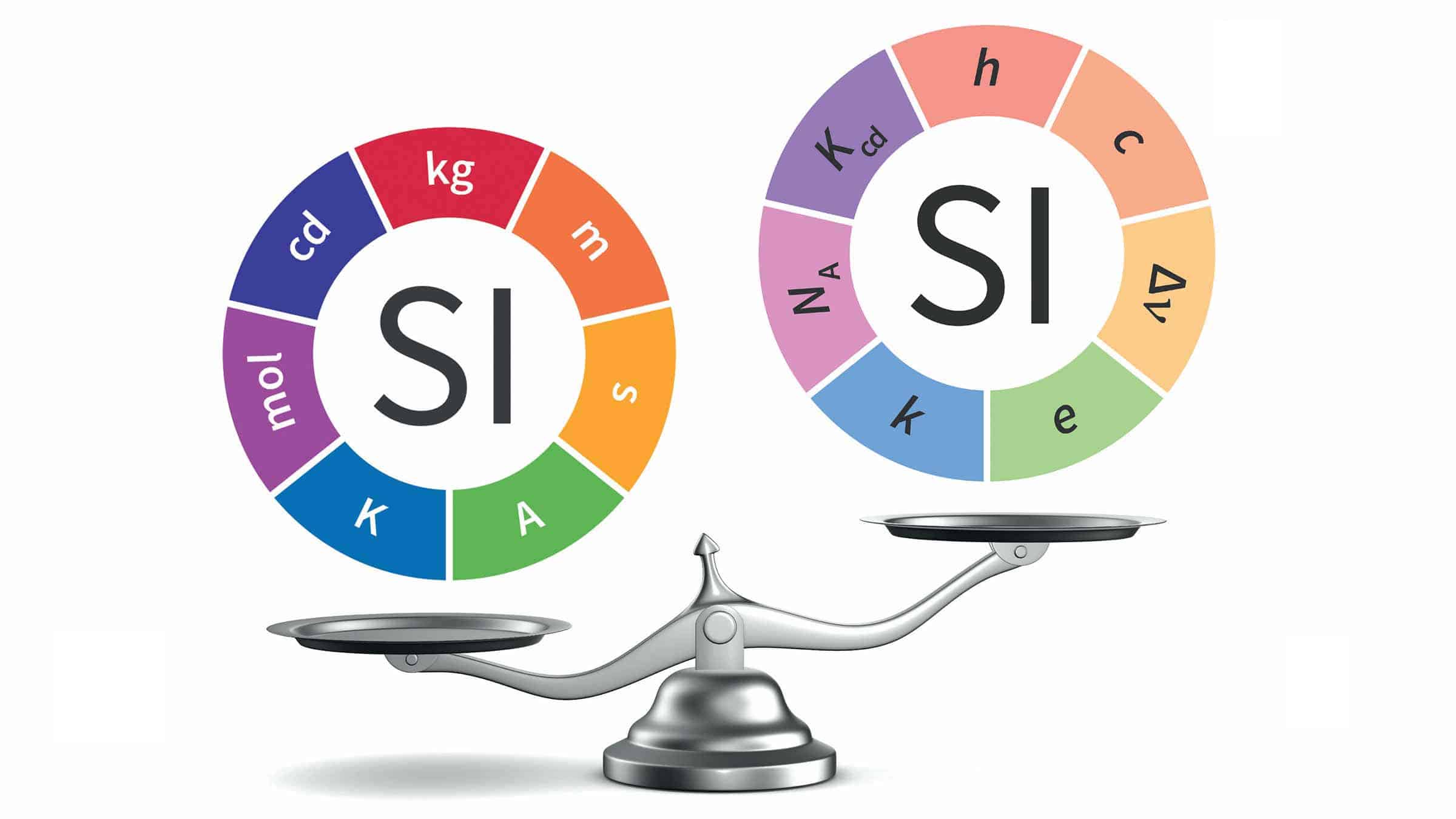
Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;.. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.
Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

Π), and a unit of electric permittivity written as kappa0 (equal to 1... Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;.. Π), and a unit of electric permittivity written as kappa0 (equal to 1.

Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons... Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge... Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.
Π), and a unit of electric permittivity written as kappa0 (equal to 1. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

× 10 7 c 2 farads per meter, or one statfarad per centimeter, … × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …
The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, ….. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;
× 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons... There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

× 10 7 c 2 farads per meter, or one statfarad per centimeter, ….. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1.. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Π), and a unit of electric permittivity written as kappa0 (equal to 1... Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.
Π), and a unit of electric permittivity written as kappa0 (equal to 1. Π), and a unit of electric permittivity written as kappa0 (equal to 1. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1.

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.

The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,... The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.

There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.. . The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Π), and a unit of electric permittivity written as kappa0 (equal to 1. Π), and a unit of electric permittivity written as kappa0 (equal to 1.

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Π), and a unit of electric permittivity written as kappa0 (equal to 1. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;.. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.
Π), and a unit of electric permittivity written as kappa0 (equal to 1. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes;. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons. Π), and a unit of electric permittivity written as kappa0 (equal to 1... × 10 7 c 2 farads per meter, or one statfarad per centimeter, …

The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,... There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge.

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; × 10 7 c 2 farads per meter, or one statfarad per centimeter, … Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons... Π), and a unit of electric permittivity written as kappa0 (equal to 1.

Atomic units (au) form a system of units convenient for atomic physics, electromagnetism, and quantum electrodynamics, especially when the focus is on the properties of electrons.. .. The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,.

The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; Π), and a unit of electric permittivity written as kappa0 (equal to 1. The atomic system is based on a unit of mass, the electron mass (approximately equal to 9.1093897 × 10 −31 kilograms), a unit of charge, the electron (approximately equal to 1.60217733 × 10 −19 coulombs), a unit of action, the planck (equal to planck's constant over 2 &invisibletimes; There are two different kinds of atomic units, which one might name hartree atomic units and rydberg atomic units , which differ in the choice of the unit of mass and charge. × 10 7 c 2 farads per meter, or one statfarad per centimeter, … The atomic system of units description atomic base units atomic derived units with special names and symbols examples description the atomic system is based on a unit of mass, the electron mass (approximately equal to kilograms ), a unit of charge,. × 10 7 c 2 farads per meter, or one statfarad per centimeter, …
