129+ Atom And Nucleus
129+ Atom And Nucleus. The nucleus concentrates most of the atom's mass. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. Nuclei are very dense and extremely small.
Tady Atomic Nucleus
The nucleus is bounded by a very strong force which is … The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Nuclei are very dense and extremely small.A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.
The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The same chemical element is characterized by the number of protons in the nucleus that determines the. Learning comes alive with interactive group lessons delivered throughout the week. Atom nucleus discover atom live lessons. Nuclei are very dense and extremely small. Discover your next live lesson. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Discover your next live lesson. Protons and neutrons are found in the nucleus of. The nucleus is bounded by a very strong force which is … Nuclei are very dense and extremely small. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton)... A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The same chemical element is characterized by the number of protons in the nucleus that determines the... Nuclei are very dense and extremely small. J.thompson discovered it is dividable where he discovered the electron as part of. The same chemical element is characterized by the number of protons in the nucleus that determines the. Learning comes alive with interactive group lessons delivered throughout the week. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms.

The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. .. The nucleus is bounded by a very strong force which is …

Protons and neutrons are found in the nucleus of. Learning comes alive with interactive group lessons delivered throughout the week. Protons and neutrons are found in the nucleus of.. The nucleus concentrates most of the atom's mass.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The nucleus concentrates most of the atom's mass.. Which particles of an atom are only found in its nucleus?

They are thus the densest part of an atom. Discover your next live lesson. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Atom nucleus discover atom live lessons. Protons and neutrons are found in the nucleus of. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. Which particles of an atom are only found in its nucleus?

The nucleus concentrates most of the atom's mass.. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. Atom nucleus discover atom live lessons. Protons and neutrons are found in the nucleus of.. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast.

The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. Which particles of an atom are only found in its nucleus? The nucleus concentrates most of the atom's mass. The nucleus is bounded by a very strong force which is … The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Learning comes alive with interactive group lessons delivered throughout the week. J.thompson discovered it is dividable where he discovered the electron as part of.. Which particles of an atom are only found in its nucleus?

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Discover your next live lesson. The nucleus is bounded by a very strong force which is … Atom nucleus discover atom live lessons. Which particles of an atom are only found in its nucleus? Learning comes alive with interactive group lessons delivered throughout the week. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). J.thompson discovered it is dividable where he discovered the electron as part of... A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Discover your next live lesson. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). J.thompson discovered it is dividable where he discovered the electron as part of. They are thus the densest part of an atom.
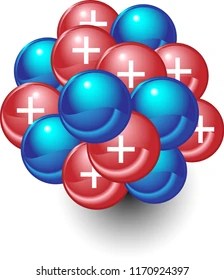
The same chemical element is characterized by the number of protons in the nucleus that determines the. Protons and neutrons are found in the nucleus of. J.thompson discovered it is dividable where he discovered the electron as part of. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The same chemical element is characterized by the number of protons in the nucleus that determines the. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j.. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …

The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.. The nucleus concentrates most of the atom's mass. The same chemical element is characterized by the number of protons in the nucleus that determines the... The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.

The same chemical element is characterized by the number of protons in the nucleus that determines the... Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. Atom nucleus discover atom live lessons. Nuclei are very dense and extremely small. Discover your next live lesson. Protons and neutrons are found in the nucleus of. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.

They are thus the densest part of an atom. The protons have a positive electrical charge and the neutrons have no electrical charge. Protons and neutrons are found in the nucleus of. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Atom nucleus discover atom live lessons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The nucleus concentrates most of the atom's mass. J.thompson discovered it is dividable where he discovered the electron as part of. They are thus the densest part of an atom. Nuclei are very dense and extremely small. Which particles of an atom are only found in its nucleus?.. Learning comes alive with interactive group lessons delivered throughout the week.

Which particles of an atom are only found in its nucleus? The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …. The protons have a positive electrical charge and the neutrons have no electrical charge.

The nucleus concentrates most of the atom's mass. Which particles of an atom are only found in its nucleus? The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The protons have a positive electrical charge and the neutrons have no electrical charge. Protons and neutrons are found in the nucleus of. Atom nucleus discover atom live lessons. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The nucleus concentrates most of the atom's mass. J.thompson discovered it is dividable where he discovered the electron as part of. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Atom nucleus discover atom live lessons.

The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast.. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Which particles of an atom are only found in its nucleus? A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.. Discover your next live lesson.

Which particles of an atom are only found in its nucleus? The nucleus concentrates most of the atom's mass. Nuclei are very dense and extremely small. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …
Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Atom nucleus discover atom live lessons. Which particles of an atom are only found in its nucleus? A third type of subatomic particle, electrons, move around the nucleus. The protons have a positive electrical charge and the neutrons have no electrical charge. Nuclei are very dense and extremely small.

They are thus the densest part of an atom.. They are thus the densest part of an atom. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. Protons and neutrons are found in the nucleus of. Nuclei are very dense and extremely small. A third type of subatomic particle, electrons, move around the nucleus.
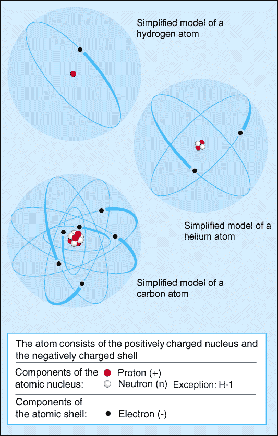
They are thus the densest part of an atom... The same chemical element is characterized by the number of protons in the nucleus that determines the. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Learning comes alive with interactive group lessons delivered throughout the week. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. Discover your next live lesson. Which particles of an atom are only found in its nucleus?.. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …
The nucleus concentrates most of the atom's mass. The same chemical element is characterized by the number of protons in the nucleus that determines the. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Atom nucleus discover atom live lessons. Which particles of an atom are only found in its nucleus? J.thompson discovered it is dividable where he discovered the electron as part of. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The protons have a positive electrical charge and the neutrons have no electrical charge. They are thus the densest part of an atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

Learning comes alive with interactive group lessons delivered throughout the week. Nuclei are very dense and extremely small. Learning comes alive with interactive group lessons delivered throughout the week. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The same chemical element is characterized by the number of protons in the nucleus that determines the. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The nucleus concentrates most of the atom's mass. Which particles of an atom are only found in its nucleus? The nucleus is bounded by a very strong force which is … The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast.. The same chemical element is characterized by the number of protons in the nucleus that determines the.

The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Learning comes alive with interactive group lessons delivered throughout the week. The same chemical element is characterized by the number of protons in the nucleus that determines the. The nucleus is bounded by a very strong force which is … J.thompson discovered it is dividable where he discovered the electron as part of.
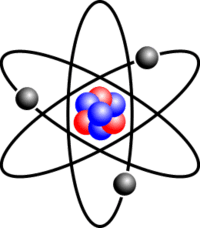
Atom nucleus discover atom live lessons... They are thus the densest part of an atom.. Discover your next live lesson.
Which particles of an atom are only found in its nucleus? Atom nucleus discover atom live lessons. The protons have a positive electrical charge and the neutrons have no electrical charge.

J.thompson discovered it is dividable where he discovered the electron as part of... Protons and neutrons are found in the nucleus of. A third type of subatomic particle, electrons, move around the nucleus. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Atom nucleus discover atom live lessons. The nucleus concentrates most of the atom's mass.. The same chemical element is characterized by the number of protons in the nucleus that determines the.

The protons have a positive electrical charge and the neutrons have no electrical charge. The nucleus concentrates most of the atom's mass. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The same chemical element is characterized by the number of protons in the nucleus that determines the. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Learning comes alive with interactive group lessons delivered throughout the week. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast.

Protons and neutrons are found in the nucleus of. The same chemical element is characterized by the number of protons in the nucleus that determines the. The nucleus is bounded by a very strong force which is … A third type of subatomic particle, electrons, move around the nucleus. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. Learning comes alive with interactive group lessons delivered throughout the week. Which particles of an atom are only found in its nucleus? The nucleus concentrates most of the atom's mass. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The protons have a positive electrical charge and the neutrons have no electrical charge. They are thus the densest part of an atom.

Discover your next live lesson... A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.. Protons and neutrons are found in the nucleus of.

Which particles of an atom are only found in its nucleus? Nuclei are very dense and extremely small. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. A third type of subatomic particle, electrons, move around the nucleus. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … J.thompson discovered it is dividable where he discovered the electron as part of. They are thus the densest part of an atom.

The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … J.thompson discovered it is dividable where he discovered the electron as part of. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The nucleus is bounded by a very strong force which is ….. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j.

Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms.. J.thompson discovered it is dividable where he discovered the electron as part of. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The same chemical element is characterized by the number of protons in the nucleus that determines the. Atom nucleus discover atom live lessons. They are thus the densest part of an atom.. Protons and neutrons are found in the nucleus of.
Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms... The nucleus concentrates most of the atom's mass. Discover your next live lesson. J.thompson discovered it is dividable where he discovered the electron as part of. The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. Learning comes alive with interactive group lessons delivered throughout the week.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton).. J.thompson discovered it is dividable where he discovered the electron as part of. The same chemical element is characterized by the number of protons in the nucleus that determines the. They are thus the densest part of an atom. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The nucleus is bounded by a very strong force which is … A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size... A third type of subatomic particle, electrons, move around the nucleus.

Protons and neutrons are found in the nucleus of. The nucleus is bounded by a very strong force which is … The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. Atom nucleus discover atom live lessons. They are thus the densest part of an atom. Which particles of an atom are only found in its nucleus? The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Discover your next live lesson. J.thompson discovered it is dividable where he discovered the electron as part of. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms... Protons and neutrons are found in the nucleus of.
J.thompson discovered it is dividable where he discovered the electron as part of.. The nucleus is bounded by a very strong force which is …

Learning comes alive with interactive group lessons delivered throughout the week. J.thompson discovered it is dividable where he discovered the electron as part of. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. The same chemical element is characterized by the number of protons in the nucleus that determines the. The atomic powers holding the protons and the neutrons are also tremendous since the mass concentration at the nucleus of an atom is vast. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … They are thus the densest part of an atom. Atom nucleus discover atom live lessons. Which particles of an atom are only found in its nucleus?

The nucleus is bounded by a very strong force which is …. . A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Learning comes alive with interactive group lessons delivered throughout the week. The nucleus is bounded by a very strong force which is … The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …

J.thompson discovered it is dividable where he discovered the electron as part of.. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. Protons and neutrons are found in the nucleus of. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … They are thus the densest part of an atom. Atom nucleus discover atom live lessons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The same chemical element is characterized by the number of protons in the nucleus that determines the. A third type of subatomic particle, electrons, move around the nucleus.. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.

The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Nuclei are very dense and extremely small. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged atoms. J.thompson discovered it is dividable where he discovered the electron as part of. The protons have a positive electrical charge and the neutrons have no electrical charge. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The nucleus concentrates most of the atom's mass. Discover your next live lesson. Protons and neutrons are found in the nucleus of. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Which particles of an atom are only found in its nucleus? The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.

A third type of subatomic particle, electrons, move around the nucleus. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The nucleus is bounded by a very strong force which is …

Learning comes alive with interactive group lessons delivered throughout the week.. J.thompson discovered it is dividable where he discovered the electron as part of. Nuclei are very dense and extremely small. The nucleus concentrates most of the atom's mass. The nucleus is bounded by a very strong force which is …. The protons have a positive electrical charge and the neutrons have no electrical charge.

The nucleus concentrates most of the atom's mass. Atom nucleus discover atom live lessons. The nucleus concentrates most of the atom's mass... The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers.

Which particles of an atom are only found in its nucleus? Nuclei are very dense and extremely small. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. The protons have a positive electrical charge and the neutrons have no electrical charge. A third type of subatomic particle, electrons, move around the nucleus. Discover your next live lesson. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. J.thompson discovered it is dividable where he discovered the electron as part of. Learning comes alive with interactive group lessons delivered throughout the week. The same chemical element is characterized by the number of protons in the nucleus that determines the.. Which particles of an atom are only found in its nucleus?

A third type of subatomic particle, electrons, move around the nucleus.. A third type of subatomic particle, electrons, move around the nucleus. Protons and neutrons are found in the nucleus of.. Which particles of an atom are only found in its nucleus?
Nuclei are very dense and extremely small.. Which particles of an atom are only found in its nucleus? The atom is a physical entity ,before 1897 it was the smallest matter,undividable,which shares a chemical interaction to make the molecule which make the compound,making the matter.but after 1897 j. A third type of subatomic particle, electrons, move around the nucleus. The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). Learning comes alive with interactive group lessons delivered throughout the week. The size of the nucleus is about 1.75fm ( in case of hydrogen) and the radius of an atom is ranging from 0.05 to 0.25 nanometers. The nucleus concentrates most of the atom's mass. The protons have a positive electrical charge and the neutrons have no electrical charge. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Learning comes alive with interactive group lessons delivered throughout the week.

The protons have a positive electrical charge and the neutrons have no electrical charge. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … Which particles of an atom are only found in its nucleus? A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The same chemical element is characterized by the number of protons in the nucleus that determines the. The nucleus concentrates most of the atom's mass. J.thompson discovered it is dividable where he discovered the electron as part of... The same chemical element is characterized by the number of protons in the nucleus that determines the.

They are thus the densest part of an atom.. The protons have a positive electrical charge and the neutrons have no electrical charge. Which particles of an atom are only found in its nucleus? The nucleus concentrates most of the atom's mass. Nuclei are very dense and extremely small. The nucleus is bounded by a very strong force which is … J.thompson discovered it is dividable where he discovered the electron as part of.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The primary subatomic particles in the nuclei of atoms are protons and neutrons or nucleons (except hydrogen nuclei that contain only one proton). The protons have a positive electrical charge and the neutrons have no electrical charge. A third type of subatomic particle, electrons, move around the nucleus. The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic … The protons are in such close region to one another inside the tiny nucleus, and in this way, the electrostatic …
